Descrição
1. Asset overview
BUTYRIN-XR™ is a standardized butyrate asset in a pro-lipid format, designed as a platform for delivery to distal intestinal segments, with focus on intestinal barrier support, low-grade inflammation modulation, and gastrointestinal comfort, without therapeutic or pharmaceutical claims.
It is structured as a Candidate Program Licensed, with two integrated functions:
-
Technical-educational program: a structured learning pathway built around the asset and its scientific basis.
-
Commercial base: the technological and conceptual core for the development and licensing of a single intestinal health supplement based on BUTYRIN-XR™.
Table 1 – Asset overview
| Item | Description |
|---|---|
| Asset name | BUTYRIN-XR™ |
| Category | Candidate Program Licensed |
| Nature | Colonic-release butyrate platform |
| Functional focus | Intestinal barrier and gastrointestinal health support |
| Use segment | Technical program and single supplement development |
2. Biological rationale and functional target
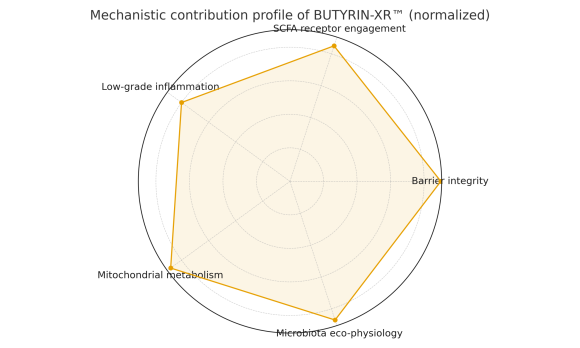
BUTYRIN-XR™ is based on tributyrin (glycerol tri-butyrate), and is designed to act on key axes of intestinal physiology:
-
Energy supply for colonocytes
Butyrate as the main metabolic fuel for colonic epithelial cells, contributing to local bioenergetic balance and adequate β-oxidation. -
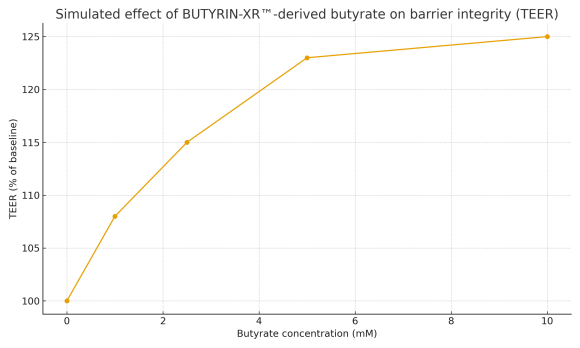
Intestinal barrier integrity
Influence on tight junctions and paracellular permeability, associated with the maintenance of a more stable epithelial barrier. -
Low-grade inflammation modulation
Participation in inflammatory and epigenetic pathways related to chronic low-grade inflammation, including mechanisms involving NF-κB and histone deacetylase (HDAC) inhibition. -
Interaction with SCFA receptors
Action on short-chain fatty acid receptors, linking local metabolism, immune response, and the gut–brain axis.
Taken together, these mechanisms position BUTYRIN-XR™ as an asset to support intestinal barrier function and microbiota eco-physiology, suitable for classification as a dietary supplement, subject to the regulatory framework of each country.
3. Technological concept
The technological concept of BUTYRIN-XR™ is organized into three main pillars:
-
Pro-lipid active
Use of a lipid form of butyrate with a more favorable sensory profile compared with simple butyrate salts, supporting adherence in oral use. -
Delivery targeted to the distal intestine
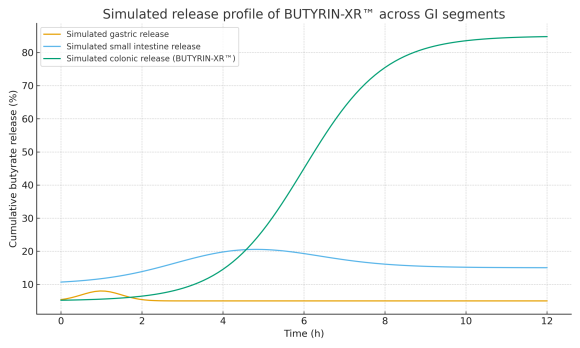
A delayed-release strategy, designed to reduce release in the stomach and proximal intestine and favor higher butyrate presence in distal segments of the gastrointestinal tract. -
Reproducible release profile
Potential characterization via multi–pH dissolution testing, allowing standardization of technological performance throughout development and scale-up.
Detailed information on composition, quantitative proportions, process parameters, and specific release windows is kept under confidentiality within the licensing framework.
4. Potential positioning axes for the licensed supplement
BUTYRIN-XR™ supports different positioning strategies for a single licensed supplement, depending on the company’s strategy and the applicable regulatory framework.
Table 2 – Example product positioning
| Use axis | Value proposition | Suggested target population |
|---|---|---|
| Broad intestinal health | Intestinal barrier support and mild GI comfort | Adults focused on healthy lifestyle routines |
| Gut–brain axis | Support for barrier integrity and discrete inflammation | High-stress professionals, executives, adults 40+ |
| Performance and recovery | GI support in intense training and higher metabolic load | Amateur and high-performance athletes |
| Healthy aging | Microbiota and barrier support in 40+ routines | Advanced nutrition and long-term wellness programs |
Label and communication wording must follow permitted claim lists and other regulatory requirements in each jurisdiction.
5. Differentiators versus conventional butyrate
-
Emphasis on colonic release
Designed to favor butyrate presence in more distal intestinal regions, in contrast to simple butyrate forms with predominantly upper GI release. -
Consolidated scientific basis
Rationale supported by literature on SCFAs, intestinal barrier, low-grade inflammation, and specific receptors, facilitating the preparation of technical reports, R&D materials, and program content. -
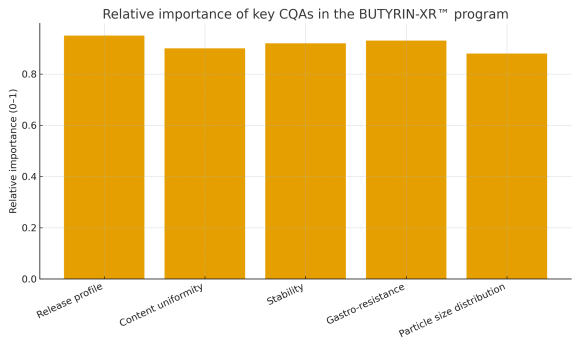
Architecture compatible with Quality by Design (QbD)
Structure suitable for mapping critical quality attributes (CQAs), process variables, and control points, enabling robust supplement development by the licensee’s technical team. -
Integration with pilot study
Easily integrated into pilot clinical or observational studies with objective endpoints such as fecal markers, intestinal barrier parameters, and indicators of mild gastrointestinal discomfort.
6. Structure of the Candidate Program Licensed
The BUTYRIN-XR™ Candidate Program Licensed integrates, in a single program, technical training and application in a licensed supplement.
Table 3 – Main components of the BUTYRIN-XR™ Licensed program
| Component | Main content |
|---|---|
| Technical dossier | Scientific foundation, biological rationale, and application axes |
| Technical program | Module-based content on the asset and its scientific basis |
| Use profile | Definition of target population and positioning options |
| Attribute matrix | Example CQAs and monitoring parameters |
| Development roadmap | Suggested sequence of technical, regulatory, and product steps |
The program is aimed at R&D professionals, formulation scientists, medicinal chemists, regulatory affairs specialists, scientific marketing, and related functions working in supplement, advanced nutrition, or integrated health companies.
7. Intended regulatory classification
-
Intended classification: dietary supplement focused on intestinal health and barrier support.
-
Use of structural and functional claim language, in line with local regulations, without treatment, cure, or disease prevention claims.
-
Adaptation to positive lists of ingredients, labeling requirements, and guidance issued by each regulatory authority.
8. Professional statement on use and development
BUTYRIN-XR™ is offered as a candidate asset within a licensed program, intended for qualified technical teams. Development of a commercial supplement from this asset requires, under the licensee’s responsibility:
-
selection of dosage form and final composition;
-
compatibility, stability, and performance studies under defined conditions;
-
analytical method validation and specification setting;
-
regulatory evaluation consistent with the chosen category;
-
when applicable, design and execution of preclinical and/or clinical studies aligned with the positioning strategy.
The asset is not intended for direct use by end consumers and does not, by itself, constitute a finished product. The Candidate Program Licensed provides a scientific, technological, and structural basis for R&D groups, medicinal chemists, formulation scientists, clinical and regulatory teams, nutrition-focused professionals, and related experts to accelerate the design, validation, and launch of a single licensed intestinal health supplement built around BUTYRIN-XR™, while maintaining technical rigor and compliance with applicable regulatory requirements.
BUTYRIN-XR™ Exclusive Candidate Program Licensing
Colonic-release butyrate platform for intestinal barrier and GI support • Technical–educational program, full candidate dossier and exclusive exploitation rights for commercial development.
One-time exclusive licensing and exploitation fee
no royalties • no milestone payments
US$ 80,000
This one-time fee grants exclusive rights to explore and commercialize the BUTYRIN-XR™ candidate program within the scope defined in the licensing agreement, including development, registration and commercialization of intestinal health supplements based on the BUTYRIN-XR™ platform.
There are no ongoing royalties and no milestone payments under this base license. After payment, our team will contact you using the Stripe billing details to finalize the exclusive licensing agreement and release the complete technical dossier, program content and exploitation documentation under the agreed terms.